Introduction
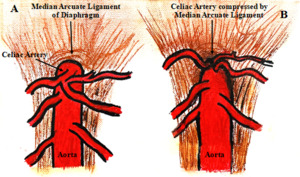
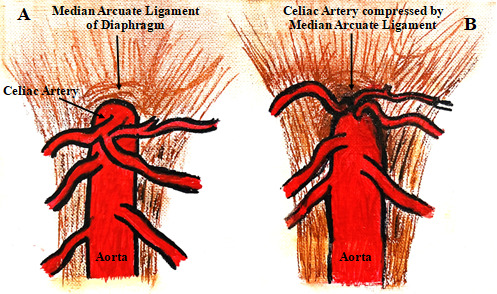
Median arcuate ligament syndrome (MALS), also known as celiac artery compression syndrome or abdominal angina, is an underdiagnosed chronic pain condition in which the median arcuate ligament compresses the celiac artery at its origin from the aorta.(Figure 1) This syndrome was first described by Lipshutz in 1917, followed by Harjola in 1963 and Dunbar in 1965.1 The median arcuate ligament, formed by the left and right diaphragmatic crura, typically passes superiorly to the origin of the celiac artery.(Figure 1A) However, in 10-24% of the population, the ligament is low-lying, which in some cases can lead to MALS.2 (Figure 1B) MALS, affecting 2 out of every 100,000 people, most commonly occurs in women aged 30 to 50 years old and is characterized by a triad of post-prandial abdominal pain, weight loss, and nausea and vomiting.2 The abdominal pain is hypothesized to result from foregut ischemia secondary to celiac artery compression, irritation of the sympathetic pain fibers of celiac ganglia, causing splanchnic vasoconstriction and further ischemia, or a combination of both mechanisms.3
Case Report
A 32-year-old female patient (BMI-22.5 kg/m2) with a history of Ehlers-Danlos syndrome (EDS) with postural orthostatic tachycardia syndrome (POTS) was scheduled for open repair of MALS under general anesthesia. The patient’s symptoms included intermittent abdominal pain after eating. Her history was significant for dysautonomia, syncope, deep vein thrombosis, anxiety, bipolar disorder, cholecystectomy, and gastric sleeve surgery three years ago. Her dysautonomia was managed on multiple medications including pyridostigmine, midodrine, droxidopa, and fludrocortisone. The patient received a celiac plexus block two months before the planned surgery, which improved her symptoms. Based on her nonspecific abdominal pain, symptomatic pain relief after a celiac plexus block, and CT abdomen angiogram, she was diagnosed with MALS.
She underwent open median arcuate ligament release and celiac plexus neurolysis with lumbar sympathectomy under general anesthesia with endotracheal intubation. Intraoperative monitoring included standard American Society of Anesthesiologists (ASA) monitors (EKG, SpO2, EtCO2, and temperature), neuromuscular monitoring, invasive blood pressure monitoring via an arterial line, and a Foley catheter. Anesthetic induction included propofol (100 mg), fentanyl (50 mcg), midazolam (2 mg), lidocaine (50 mg), and rocuronium (50 mg). Anesthesia was maintained with sevoflurane in an oxygen-air mixture, with titrated doses of fentanyl for analgesia, rocuronium for neuromuscular blockade, and ondansetron for prophylaxis of postoperative nausea and vomiting. At the conclusion of surgery, neuromuscular blockade was reversed with sugammadex. The patient was extubated in the operating room and transferred to the post-anesthesia care unit (PACU) without intraoperative complications.
For enhanced postoperative pain control, a thoracic epidural catheter was placed preoperatively at the T8-T9 level and utilized for three days postoperatively, providing adequate analgesia. The postoperative course was notable for mild nausea and vomiting, which was effectively treated with ondansetron as needed. The patient recovered and was discharged home. At a two-week follow-up visit in the vascular surgery clinic, she reported maximal symptomatic improvement following surgery.
Discussion
Preoperative Preparation and Workup
MALS symptoms overlap with numerous gastrointestinal disorders, making the diagnosis of patients particularly challenging. Timely diagnosis is critical, as undiagnosed patients may undergo unnecessary procedures, such as diagnostic laparoscopy, cholecystectomy, and laparotomy, and continue to experience unmanaged chronic abdominal pain.4 Positive workup reveals elevated celiac artery velocities on ultrasound that normalize with inspiration and a ‘hooked’ configuration of the celiac artery on CT angiography.2 MALS remains a diagnosis of exclusion, and alternative etiologies of abdominal pain must be carefully ruled out, particularly given that many individuals with celiac artery compression by the median arcuate ligament are asymptomatic.1
Several conditions can occur comorbidly with MALS, including dysautonomia such as POTS, connective tissue disorders such as EDS, and psychiatric conditions. These comorbid conditions should be carefully considered in surgical decision-making due to their potential impact on outcomes. The mainstay treatment for MALS is surgical release of the median arcuate ligament. However, in patients with both MALS and POTS, as in this case, concomitant celiac plexus sympathectomy should be considered, as symptomatology may reflect a greater neuropathic component.¹ POTS is characterized by systemic autonomic dysregulation with prominent gastrointestinal manifestations, which may predispose patients to exaggerated neurogenic abdominal pain when superimposed upon direct celiac plexus irritation from median arcuate ligament compression.5
Preoperative evaluation of a patient’s medications is also essential. This evaluation is crucial for patients with dysautonomia, as treatment aimed at optimizing hemodynamics may interact with anesthesia, thereby heightening the risk of intraoperative complications. Several medications frequently prescribed for POTS necessitate careful consideration.(Table 1)
-
Pyridostigmine is an acetylcholinesterase inhibitor that enhances baroreflex function, helping to reduce excessive tachycardia. This inhibitor can cause unpredictable neuromuscular blockade with agents like succinylcholine due to competition at nicotinic receptors.6 Therefore, careful quantitative neuromuscular blockade monitoring with a peripheral nerve stimulator is essential to assess the degree of paralysis and guide neuromuscular blockade dosing. Pyridostigmine can also alter responsiveness to reversal agents such as neostigmine. Their combined cholinergic effects may potentiate bradycardia. Withholding of pyridostigmine in the preoperative period is generally safe and should be considered by anesthesiologists in patients with severe autonomic dysfunction, such as autonomic failure.6 The amount of time to stop pyridostigmine before surgery is often individualized based on the patient’s symptoms, surgery type, and anesthetic plan.
-
Midodrine, an alpha-1 adrenergic agonist, induces vasoconstriction, improving venous return, blood pressure, and cerebral perfusion, thereby reducing the orthostatic tachycardia, dizziness, and fatigue seen in POTS. However, this agonist can cause supine hypertension intraoperatively, which may be managed with reverse Trendelenburg positioning.6 Anesthesiologists should consider holding the drug on the morning of surgery, especially in patients with preexisting supine hypertension.6
-
Fludrocortisone is a mineralocorticoid that promotes renal sodium retention and water reabsorption, helping to reduce orthostatic tachycardia. It can cause hypokalemia in up to 50% of patients and hypomagnesemia in up to 10%, increasing the risk of arrhythmias and altered response to neuromuscular blockers.6 Doses above 0.3 mg/day may suppress the hypothalamic-pituitary-adrenal (HPA) axis, impairing the cortisol stress response to surgery.6 If suppression is present, stress-dose steroids may be required to maintain hemodynamic stability.
A thorough review of a patient’s medications informs anesthetic choices and enhances preparedness to manage potential perioperative complications.
Surgical and Anesthetic Considerations
Patient selection is the most important determinant of successful surgical outcomes.1 Recommendation for surgery should be determined by a multidisciplinary team of vascular surgery, pain medicine, and psychiatry.1 Certain factors increase the likelihood of persistent symptoms even after the surgery. These factors include atypical pain, periods of remission, age over 60 years, a history of psychiatric conditions or alcohol abuse, weight loss of less than 10 kg, and dysautonomia.1 In patients with these risk factors who still opt for surgery, it is important to manage expectations and emphasize that it may not be curative.1 Conversely, post-prandial pain, age between 40 and 60 years, and weight loss of 10 kg or greater are associated with more successful outcomes.1
A celiac plexus block with local anesthetic by a pain physician may help to determine both the diagnostic and therapeutic approach for MALS. Surgical treatment for MALS involves laparoscopic, open, or robotic release of the median arcuate ligament, with reported success rates of 70% to 80%.1 This procedure may be combined with celiac plexus neurolysis, revascularization, or both, depending on the individual patient. Intraoperative confirmation of successful release can be achieved using duplex ultrasound, which demonstrates minimal change in celiac artery velocity at baseline and during deep inspiration, as described by Skelly and Mak at the University of Chicago.1 Laparoscopic surgery is generally favored over open approaches because it offers smaller incisions, reduced postoperative morbidity, and faster recovery.2 However, laparoscopy is also associated with a higher risk of aortic injury, incomplete ligament release, and difficult-to-control hemorrhage.2 If an aortic injury occurs, conversion to emergent open surgery is required to minimize morbidity and mortality. For this reason, anesthesiologists should be prepared for the possibility of major blood loss and ensure that blood products are readily available.7
Intraoperative Complications
Hypotension is a concern during the release of the median arcuate ligament because hemodynamic lability is common in patients with POTS. Impaired autonomic vasoconstriction and relative hypovolemia increase their susceptibility to blood pressure fluctuations under anesthesia. Hypotension can be managed with crystalloid fluid boluses.8 If blood pressure does not improve with fluid resuscitation, phenylephrine should be used as the first-line agent due to its selective alpha-1 adrenergic activity, which promotes arterial and venous vasoconstriction and thereby increases blood pressure.8 Epidural analgesia can improve postoperative pain control in patients with MALS undergoing release of the median arcuate ligament, but it is contraindicated in those with preexisting hypovolemia due to the risk of neuraxial-induced hypotension.8 Because patients with POTS are often relatively hypovolemic and particularly susceptible to hypotension under anesthesia, ensuring adequate intravenous hydration before epidural administration is essential to minimize this risk.8
Compression of the celiac artery in MALS reduces blood flow to the gastrointestinal system. The resulting chronic ischemia slows gastric emptying and impairs gastrointestinal motility, producing the classic symptoms of post-prandial abdominal pain, weight loss, nausea, and vomiting. In patients with comorbid dysautonomia, such as POTS, these symptoms may be exacerbated due to further impairment of gastrointestinal motility. For anesthesiologists, gastroparesis carries significant implications for airway management, as it increases the risk of aspiration. Rapid sequence induction and intubation should be considered in these patients.6
Anesthesiologists must recognize that patients with dysautonomia are sensitive to positive pressure ventilation and pneumoperitoneum, the latter of which is required for laparoscopic procedures like median arcuate ligament release.6 These interventions reduce venous return to the heart. In healthy individuals, the sympathetic nervous system compensates by vasoconstricting and modestly increasing heart rate. POTS patients rely primarily on heart rate to maintain cardiac output due to impaired vascular compensation, often resulting in exaggerated tachycardia and hypotension. Anesthesiologists should maintain adequate intravascular volume and consider using smaller tidal volumes to minimize hemodynamic instability.6
Postoperative Pain Management
Because MALS is a chronic pain condition, postoperative pain is often difficult to manage and typically necessitates greater care and medication requirements than for the average surgical patient. Chronic pain patients are frequently on long-term opioid therapy, which can lead to opioid-induced hyperalgesia and tolerance.9 To reduce postoperative pain after laparoscopic procedures such as median arcuate ligament release, studies recommend several strategies: injecting long-acting local anesthetics at port sites, removing residual CO₂, administering intraperitoneal subdiaphragmatic local anesthetic, and, in some cases, using epidural analgesia.2 Epidural analgesia has been shown to accelerate the return of normal gastrointestinal motility and reduce pain with movement during the first 24 hours after surgery.2 For patients who cannot receive an epidural, a combination of continuous opioid infusion with patient-controlled anesthesia (PCA) bolus doses offers a good alternative.2 Although narcotic use may worsen pre-existing gastrointestinal motility issues, pain control should not be compromised by overly minimizing their use.6
Long-Term Outcomes and Follow-Up
Although surgical release of the median arcuate ligament has a high success rate, all patients who undergo surgery must be followed long-term due to the possibility of persistent or recurrent abdominal pain.1 This pain may result from restenosis, indicated by reduced celiac artery flow, or from neuropathy if celiac artery velocity is normal, in which case a celiac plexus block may be beneficial.1 If pain persists despite normalized celiac artery velocity, absence of neuropathic features, or other biochemical abnormalities, it is likely due to a functional gastrointestinal disorder (FGID).1 In such cases, medical management by a multidisciplinary team including gastroenterology, pain physicians, vascular surgery, and psychiatry is recommended.10 For most patients, however, surgical release provides at least partial symptom relief and patient satisfaction.1
Conclusion
Physicians should maintain a high index of suspicion for MALS when a patient presents with the characteristic symptom triad of post-prandial abdominal pain, weight loss, and nausea and vomiting. In patients with MALS and coexisting POTS undergoing laparoscopic ligament release or open surgery, anesthetic care requires careful selection of agents due to potential interactions with POTS medications and thorough preparation for procedural complications. Recognizing MALS and its association with dysautonomia, such as POTS, through a multi-disciplinary approach can accelerate the diagnosis, guide treatment strategies, and ultimately improve patient outcomes.